Vapour Permeability and Deuterium: Importance, Uses and Applications
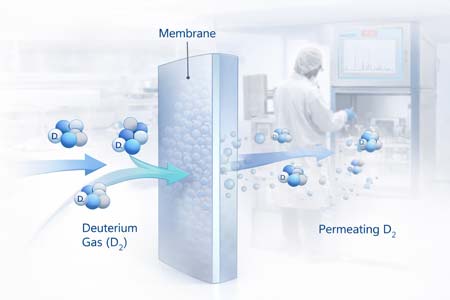 Vapour permeability describes how readily a gas or vapour can pass through a solid material such as a polymer, metal, coating, or membrane and it is a defining property in the containment, transport and separation of deuterium.
Vapour permeability describes how readily a gas or vapour can pass through a solid material such as a polymer, metal, coating, or membrane and it is a defining property in the containment, transport and separation of deuterium.
Deuterium, present as molecular deuterium (D₂) or in compounds such as heavy water (D₂O), is a strategically important isotope with applications spanning nuclear fusion research, heavy water production for nuclear reactors, advanced energy systems and materials science. The global deuterium and heavy water market represents multi-billion -pound investment in infrastructure, fuel handling and long-term supply chains, with demand accelerating as fusion programmes move from research into deployment.
In this context, accurate vapour permeability measurement is essential for material qualification, safety assurance and process optimisation and Versaperm vapour permeability measurement equipment plays a key role in delivering the precision required for high - value deuterium applications.
What Vapour Permeability Means for Deuterium
- Vapour permeability quantifies how easily deuterium can diffuse through a solid barrier.
- It applies to:
- Containment materials such as metals, alloys, polymers and ceramics
- Selective membranes used in isotope separation
- Protective coatings, liners and seals
- Vapour permeability depends on:
- Diffusion rate of the gas
- Solubility of deuterium in the material
- Temperature, pressure and microstructural properties
Uses of Vapour Permeability in Deuterium Applications
1. Nuclear Fusion Research
- Deuterium is a primary fuel candidate for fusion reactors, commonly paired with tritium.
- Vapour permeability data is critical to:
- Prevent deuterium loss through reactor walls
- Predict diffusion in plasma - facing components
- Manage isotope inventories and separation (D vs T)
- Low - permeability materials improve fuel efficiency and reactor performance.
2. Isotope Separation and Enrichment
- Selective membranes exploit small permeability differences between hydrogen isotopes.
- Vapour permeability measurements inform membrane design for:
- Heavy water (D₂O) production
- Deuterium enrichment and purification
- Accurate data underpins both technical feasibility and economic efficiency.
3. Fuel Cells and Electrolysers
Fuel Cells and Electrolysers
- Proton - exchange membranes transport hydrogen isotopes, including deuterium.
- Vapour permeability influences:
- Isotope transport behaviour
- Durability testing of membranes under isotope loading
- Deuterium is frequently used to study hydrogen transport mechanisms.
4. Cryogenic Storage and Handling
- Deuterium is commonly stored at high pressure and low temperature.
- Vapour permeability determines:
- Leak rates over time
- Material compatibility
- Long - term storage stability
- Even very low permeability values can lead to significant cumulative losses.
5. Materials Science and Safety
- Vapour permeability data supports:
- Leak prevention in laboratory and industrial settings
- Assessment of hydrogen embrittlement risks
- Prediction of long - term material degradation
- Deuterium is widely used as a tracer isotope for hydrogen transport studies.
Why Vapour Permeability is Especially Important for Deuterium
- Containment: Minimises loss of a costly and strategically important isotope.
- Efficiency: Reduces waste in fusion, separation and electrochemical systems.
- Safety: Limits risks associated with leaks, flammability and asphyxiation.
- Material Selection: Enables informed choice of polymers, metals and ceramics.
- Fundamental Research: Supports accurate modelling of hydrogen behaviour using deuterium.
Deuterium vs Protium: Key Permeability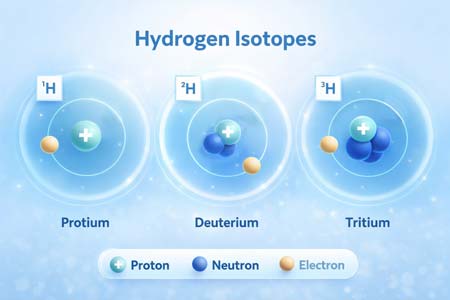 Differences
Differences
Molecular Mass and Diffusion
- Protium (H₂): ~2 g/mol
- Deuterium (D₂): ~4 g/mol
- Diffusion follows Graham’s Law:
- Heavier molecules diffuse more slowly
- :Result:
- D₂ diffuses approximately √2 times more slowly than H₂
Solubility Effects
- Both isotopes dissolve in metals and polymers.
- D₂ may show slightly higher solubility in some metals due to isotope effects.
- This can lead to greater accumulation despite slower diffusion.
Permeability Trends
- Permeability = diffusivity × solubility
- In most materials:
- H₂ permeates faster than D₂
- Typical ratios range from 1.2 to 2×
- These differences are exploited in isotope separation technologies.
 Material - Specific Behaviour
Material - Specific Behaviour
- Metals:
- Both isotopes cause embrittlement; D₂ penetrates more slowly but behaves similarly once absorbed.
- Polymers:
- Higher permeability overall; isotope effects enable selective permeation.
- Ceramics:
- Low permeability for both isotopes; essential for fusion environments.
Conclusion: Accuracy in a High-Value Deuterium Landscape
As the deuterium market continues to expand across fusion energy programmes, nuclear infrastructure and advanced materials science, the importance of reliable vapour permeability data becomes ever more pronounced. Even small inaccuracies in permeability measurement can translate into significant material losses, increased safety risks and sub-optimal material selection that ultimately shortens system lifetime and degrades performance.
Versaperm vapour permeability measurement equipment delivers the level of accuracy required to characterise materials with confidence, enabling safe containment, effective isotope management and sound engineering decisions. In deuterium applications, precise vapour permeability measurement is therefore not merely a supporting parameter but a fundamental requirement for achieving high performance, safety and long-term economic viability.


 Vapour permeability describes how readily a gas or vapour can pass through a solid material such as a polymer, metal, coating, or membrane and it is a defining property in the containment, transport and separation of deuterium.
Vapour permeability describes how readily a gas or vapour can pass through a solid material such as a polymer, metal, coating, or membrane and it is a defining property in the containment, transport and separation of deuterium.  Fuel Cells and Electrolysers
Fuel Cells and Electrolysers Differences
Differences Material - Specific Behaviour
Material - Specific Behaviour